De Novo Class II submission planned in Neurology for surgical nerve visualization.
From laboratory validation to multi-site human feasibility studies.
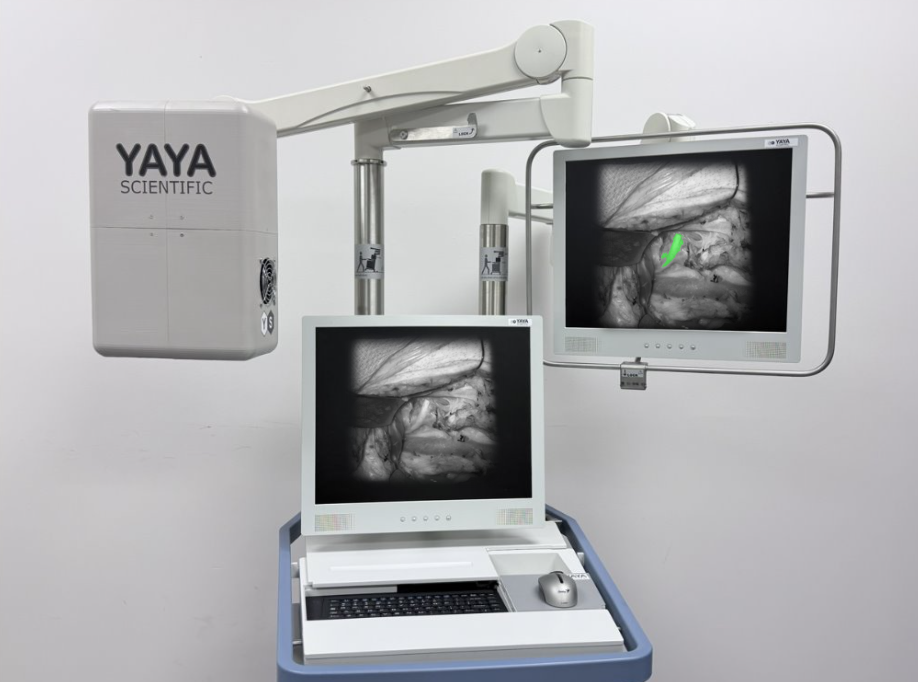
Alina™ Beta design integrating surgeon feedback into manufacturable clinical units.
Clinical Prototype and clinical study launch to support FDA clearance.
Chronic postoperative pain and nerve injury remain significant challenges across many surgical specialties in the United States. Even when overall risk is low, the consequences of nerve damage can be profound—affecting sensation, motor function, recovery time, quality of life, and long-term healthcare costs.
YaYa Scientific focuses on generating data that supports safer, more confident surgery. In collaboration with leading academic medical centers, we are evaluating whether real-time, intraoperative nerve visualization can reduce preventable injury and improve patient outcomes—without disrupting existing surgical workflows.
We study how the technology fits into standard operating room workflows—assessing usability and surgeon adoption in live cases without adding procedural complexity.
Studies examine whether real-time visualization enhances intraoperative awareness and helps surgeons identify, preserve, or repair nerves more confidently.
Our multi-site clinical studies measure safety, performance, and patient-reported outcomes to quantify the benefit of nerve visualization during surgery.
De Novo is a FDA approval pathway for novel, low-to-moderate-risk medical devices that do not have a direct predicate. When granted, it establishes the first classification for that type of device in Class II with defined special controls.
Timelines vary with device complexity, required evidence, and FDA interactions.
As YaYa Scientific progresses from Alina™ Beta verification and GLP testing to surgeon studies. This data — combined with risk analysis and manufacturing controls — form the foundation of our De Novo - Class II FDA submission.
Interested in learning more about Alina™ or collaborating with our research and clinical teams? Reach out — we’d love to hear from you.
Contact Our Team